Strata Trial
An Observational Study Profiling Biospecimens from Cancer Patients to Screen for Molecular Alterations

Cancer patients and their providers are often unaware there may be opportunities for eligibility assessments for potentially life-saving genomically-guided clinical trials and approved targeted therapies. Strata Oncology is initiating the Strata Trial (STR-001-001) with the primary goal of understanding the proportion of subjects available for clinical trials and approved targeted therapies in advanced and/or aggressive cancers while assessing the feasibility of using a large-scale NGS screening program to match subjects for eligibility assessments in clinical trials and/or for approved targeted therapies. The Strata Trial does not require additional procedures but rather uses surplus, or leftover tumor specimens for molecular profiling.
This study will plan to streamline screening for all of the affiliated therapeutic protocols simultaneously, providing clinical research options to the clinicians, tailored to the genetic alterations identified in the subjects surplus tumor specimen. The Strata Trial will also explore the effects of genomic sequencing on subject treatment selection, including standard of care regimens, clinical trial enrollment, and usage of targeted therapies.
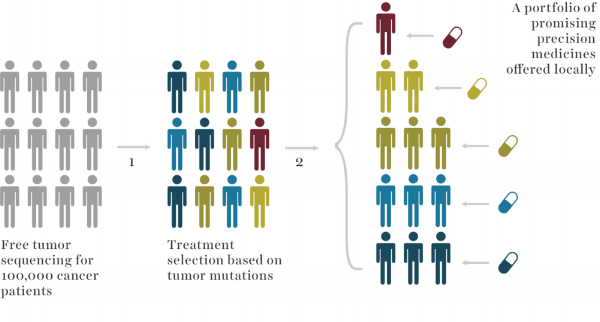
What is unique about the Strata Trial?
Cost:
- Sequencing is included at no-cost to patient, provider, or payer
Scale:
- Strata Oncology hopes to provide free screening for 10,000’s patients across 50+ institutions.
Access:
- Eligible to all advanced stage solid tumor and lymphoma patients (* additional eligibility requirements)
- Sequencing is available during standard treatment so future trial options are clear
- Readily available archival tissue is accepted for sequencing to minimize additional biopsies
Purpose:
- Most patients never know if they are eligible for a precision medicine trial
- Strata aims to substantially broaden access to tumor sequencing and precision medicine trials
- This will accelerate breakthrough trials and reduce the time to new approvals
Contact
To learn more about the program, please contact Alex Boland at aboland@med.unc.edu or (919) 966-1770.
